Three organisational architectures, three transformation logics
Why an invariants-based reading
An invariants-based approach provides a transversal analytical framework that makes it possible to compare very different organizations independently of their formal structures, organizational models, and prevailing reference frameworks.
Benchmark synthesis
This benchmark highlights three fundamentally distinct transformation logics.
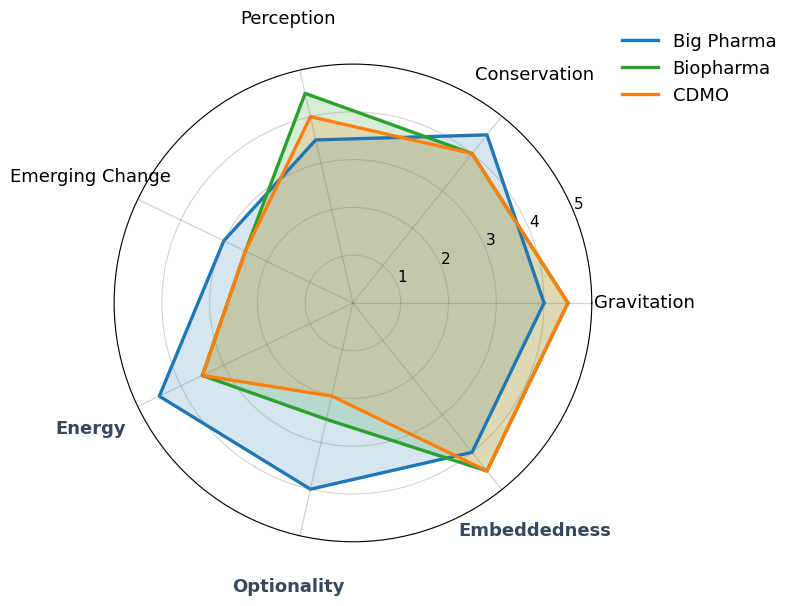
For a CDMO Contract Development and Manufacturing Organization – Manufactures complex medicines at industrial scale on behalf of pharmaceutical companies that cannot internalize this level of production complexity. , transformation primarily consists of governing industrial complexity embedded in long-lived assets and multi-client commitments, with innovation serving mainly to stabilize capacity, compliance, and execution reliability rather than to alter strategic positioning.
For an integrated Big Pharma player Discovers, develops, manufactures, and commercializes innovative medicines while controlling the full value chain from science to market. , transformation mainly aims to preserve systemic coherence across scientific, economic, and institutional layers, where data and digital capabilities act as instruments of orchestration to prevent fragmentation rather than to drive disruption.
For a specialized Biopharma player Develops and produces critical biological therapies based on rare biological systems (e.g. plasma) for severe and narrowly defined diseases. , transformation is largely about optimizing a biologically constrained system for resilience and yield, with any attempt at broad organizational reinvention being structurally unnecessary or even counterproductive.
| Invariant | CDMO | Integrated Big Pharma | Specialized Biopharma |
|---|---|---|---|
| Gravitation | Critical industrial infrastructure: without it, a portion of innovation never reaches scale | Gravitation driven by organizational mass and portfolio breadth rather than a single function | Quasi-systemic actor for certain life-critical pathologies |
| Conservation | Heavy assets, GMP certification, reliability culture → strong, accepted inertia | Very high inertia: pipelines, brands, organizations, regulators | Biological dependency (plasma), dedicated infrastructure, unique supply chains |
| Perception | Clear awareness of constraints: operational excellence > product ambition | Permanent internal tension between science, finance, narrative, and governance | Exceptional alignment between science, industry, and economic model |
| Emerging Change | Incremental, capacity-driven change; never narrative or role-based | Change is possible but slow, often absorbed by structural complexity | Structural change strongly limited by biology and logistics |
| Degree of Freedom | CDMO | Integrated Big Pharma | Specialized Biopharma |
|---|---|---|---|
| Energy | High investment capacity, tightly focused on specific industrial bets | Very high financial and scientific energy, diluted by organizational complexity | Strong investment capacity, concentrated on few strategic axes |
| Optionality | Few viable trajectories outside premium CDMO positioning | High apparent optionality (therapeutic areas, partnerships, M&A) | Expansion possible, but always adjacent to the biological core |
| Embeddedness | Deeply embedded in global regulatory and industrial value chains | Strongly embedded in healthcare systems, states, and payers | Extremely embedded (donors, regulators, states, patients) |